Detect PSMA-positive disease. Direct better care.1
PSMA is a key biomarker used to detect metastatic prostate cancer. As precision medicine grows, it supports more biomarker-guided approaches.
When you include a PSMA-PET scan as a crucial step in your PSMA plan, you can better inform decisions for your patients.
About PSMA
PSMA is a critical prostate cancer biomarker with a dual purpose1,2: | |
Detect diseasePSMA is a biomarker used to detect metastatic prostate cancer and aids in staging. It supports a more accurate treatment approach. | |
Direct patient careIncorporating PSMA imaging lets you see if, and where, prostate cancer has spread, so you can make an informed plan for your patient. | |
PSMA is a biomarker that is overexpressed in nearly all prostate cancers, regardless of stage3
Early PSMA expression detection can help inform how a patient’s prostate cancer might be managed1
About PSMA-PET Scans
PSMA-PET scans are2,4: | ||
A type of noninvasive diagnostic imaging test that uses a radiolabeled PSMA ligand to detect PSMA-positive prostate cancer cells* | ||
A potentially more effective frontline imaging tool than conventional imaging (CT, MRI, and bone scans) | ||
More sensitive and specific than conventional imaging to identify PSMA-positive lesions | ||
*PSMA can also be found on normal cells.
Compared with conventional imaging, PSMA-PET scans can provide5-7:
Superior lesion detection
More accurate staging and restaging
Changes to management of primary and recurrent prostate cancers
PSMA-PET scanning is becoming an ideal image modality to diagnose, stage, and restage PSMA-positive prostate cancer2,5,6
Practice Guidelines
NATIONAL COMPREHENSIVE CANCER NETWORK® (NCCN®) RECOMMENDED
INITIAL STAGING | |||
BIOCHEMICAL RECURRENCE | |||
AT ANY TIME TO ASSESS PROGRESSION* | |||
*With bone scan plus CT or MRI for the evaluation of bone, pelvis, and abdomen.
NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
Clinical Evidence
In clinical trials, PSMA-PET scans helped patients with advanced prostate cancer receive the following: |
MORE ACCURATE STAGING5,6,8
After a PSMA-PET scan,
were upstaged from M0 to M1
Phase 3 clinical trial of men with biochemically recurrent prostate cancer (N=1960)5,8
38.8% (n=762/1960) had metastases discovered by a PSMA-PET scan and were upstaged from M0 to M1
OSPREY: Phase 2/3 clinical trial of PSMA-PET scans in men with suspected recurrent or metastatic prostate cancer (N=385)6
57.6% (n=19/33) were upstaged to M1 after a PSMA-PET scan
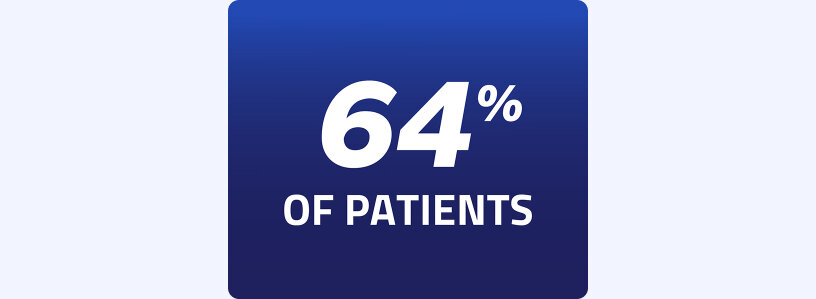
MORE ACCURATE DISEASE CLASSIFICATION9
After a PSMA-PET scan,
had a disease reclassification (n=27/67)
CHAARTED to PSMA PET: International, multicenter, retrospective study of PSMA-PET scans vs conventional imaging in men with mHSPC (N=67)9
22% (n=11/50) with low-volume disease by conventional imaging were reclassified as having high-volume disease after a PSMA-PET scan*
*High-volume disease is defined as the presence of visceral metastases or at least 4 bone metastases (≥1 beyond the vertebral bodies or the pelvis).
CHANGED MANAGEMENT PLANS7
After a PSMA-PET scan,
had a change in intended disease management plan (n=131/205)
CONDOR: Phase 3 clinical trial of PSMA-PET scans in men with biochemically recurrent prostate cancer (N=208)7
Changes in systemic therapy should not be made solely based on a positive PSMA-PET in patients with M0 CRPC.
Plan for PSMA, a dual-purpose biomarker, to1: | |
Detect disease | Direct patient care |
How can a PSMA plan
GUIDE YOUR APPROACH TO PROSTATE CANCER?
CT, computed tomography; mHSPC, metastatic hormone-sensitive prostate cancer; MRI, magnetic resonance imaging; PET, positron emission tomography; PSMA, prostate-specific membrane antigen.
1. Hupe MC, Philippi C, Roth D, et al. Front Oncol. 2018;8:623. doi:10.3389/fonc.2018.00623 2. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Prostate Cancer V.5.2026. © National Comprehensive Cancer Network, Inc. 2026. All rights reserved. Accessed January 23, 2026. To view the most recent and complete version of the guideline, go online to NCCN.org. 3. Pomykala KL, Czernin J, Grogan TR, Armstrong WR, Williams J, Calais J. J Nucl Med. 2020;61(3):405-411. doi:10.2967/jnumed.119.230318 4. Fendler WP, Eiber M, Beheshti M, et al. Eur J Nucl Med Mol Imaging. 2023;50(5):1466-1486. doi:10.1007/s00259-022-06089-w 5. Ferdinandus J, Fendler WP, Farolfi A, et al. J Nucl Med. 2022;63(1):76-80. doi:10.2967/jnumed.121.262821 6. Pienta KJ, Gorin MA, Rowe SP, et al. J Urol. 2021;206(1):52-61. doi:10.1097/JU.0000000000001698 7. Morris MJ, Rowe SP, Gorin MA, et al. Clin Cancer Res. 2021;27(13):3674-3682. doi:10.1158/1078-0432.CCR-20-4573 8. ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT02940262. Accessed December 22, 2025. 9. Unterrainer LM, Hope TA, Fendler WP, et al. J Nucl Med. 2025;66(1):54-60. doi:10.2967/jnumed.124.268441